CONSTRUCTION MATERIALS
(Source: Standard Handbook for Civil Engineers)
This section describes the basic properties of materials commonly used in construction. For convenience, materials are grouped in the following categories: cementitious materials, metals, organic materials, and composites. Application of these materials is discussed in following sections. In these sections also, environmental degradation on the materials are described.
Cementitious Materials
Any substance that bonds materials may be considered a cement. There are many types of cements. In construction, however, the term cement generally refers to bonding agents that are mixed with water or other liquid, or both, to produce a cementing paste. Initially, a mass of particles coated with the paste is in a plastic state and may be formed, or molded, into various shapes. Such a mixture may be considered a cementitious material because it can bond other materials together. After a time, due to chemical reactions, the paste sets and themass hardens. When the particles consist of fine aggregate (sand), mortar is formed. When the particles consist of fine and coarse aggregates, concrete results.
1. Types of Cementitious Materials
Cementitious materials may be classified in several different ways. One way often used is by the chemical constituent responsible for setting or hardening the cement. Silicate and aluminate cements, in which the setting agents are calcium silicates and aluminates, are the most widely used types.
Limes, wherein the hardening is due to the conversion of hydroxides to carbonates, were formerly widely used as the sole cementitious material, but their slow setting and hardening are not compatible with modern requirements. Hence, their principal function today is to plasticize the otherwise harsh cements and add resilience to mortars and stuccoes. Use of limes is beneficial in that their slow setting promotes healing, the recementing of hairline cracks.
Another class of cements is composed of calcined gypsum and its related products. The gypsum cements are widely used in interior plaster and for fabrication of boards and blocks; but the solubility of gypsum prevents its use in construction exposed to any but extremely dry climates.
Oxychloride cements constitute a class of specialty cements of unusual properties. Their cost prohibits their general use in competition with the cheaper cements; but for special uses, such as the production of sparkproof floors, they cannot be equaled.
Masonry cements or mortar cements are widely used because of their convenience. While they are, in general, mixtures of one or more of the abovementioned cements with some admixtures, they deserve special consideration because of their economies.
Other cementitious materials, such as polymers, fly ash, and silica fume, may be used as a cement replacement in concrete. Polymers are plastics with long-chain molecules. Concretes made with them have many qualities much superior to those of ordinary concrete.
Silica fume, also known asmicrosilica, is awaste product of electric-arc furnaces. The silica reacts with lime in concrete to form a cementitious material. A fume particle has a diameter only 1% of that of a cement particle.
2. Portland Cements
Particles that become a bonding agent when mixed with water are referred to as hydraulic cements. The most widely used cements in construction are portland cements, which are made by blending a mixture of calcareous (lime-containing) materials and argillaceous (clayey) materials. (See Art. 5.3 for descriptions of other types of hydraulic cements.) The raw materials are carefully proportioned to provide the desired amounts of lime, silica, aluminum oxide, and iron oxide. After grinding to facilitate burning, the raw materials are fed into a long rotary kiln, which is maintained at a temperature around 2700 °F. The raw materials, burned together, react chemically to form hard, walnut-sized pellets of a new material, clinker.
The clinker, after discharge from the kiln and cooling, is ground to a fine powder (not less than 1600 cm2/g specific surface). During this grinding process, a retarder (usually a few percent of gypsum) is added to control the rate of setting when the cement is eventually hydrated. The resulting fine powder is portland cement.
Four compounds, however, make up more than 90% of portland cement, by weight; tricalcium silicate (C3S), dicalcium silicate (C2S), tricalcium aluminate (C3A), and tetracalcium aluminoferrite (C4AF). Each of these four compounds is identifiable in the highly magnified microstructure of portland cement clinker, and each has characteristic properties that it contributes to the final mixture.
2.1 Hydration of Cement
When water is added to portland cement, the basic compounds present are transformed to new compounds by chemical reactions [Eq. (5.1)].
Two calcium silicates, which constitute about 75% of portland cement by weight, react with the water to produce two new compounds: tobermorite gel, which is not crystalline, and calcium hydroxide, which is crystalline. In fully hydrated portland cement paste, the calcium hydroxide accounts for 25% of the weight and the tobermorite gel makes up about 50%. The third and fourth
reactions in Eq. (5.1) show how the other two major compounds in portland cement combine with
water to form reaction products. The final reaction involves gypsum, the compound added to portland cement during grinding of the clinker to control set.
Each product of the hydration reaction plays a role in the mechanical behavior of the hardened paste. The most important of these, by far, is the tobermorite gel, which is the main cementing component of cement paste. This gel has a composition and structure similar to those of a naturally occurring mineral, called tobermorite, named for the area where it was discovered, Tobermory in Scotland. The gel is an extremely finely divided substance with a coherent structure.
The average diameter of a grain of portland cement as ground from the clinker is about 10 mm. The particles of the hydration product, tobermorite gel, are on the order of a thousandth of that size. Particles of such small size can be observed only by using the magnification available in an electron microscope. The enormous surface area of the gel (about 3 million cm2/g) results in attractive forces between particles since atoms on each surface are attempting to complete their unsaturated bonds by adsorption. These forces cause particles of tobermorite gel to adhere to each other and to other particles introduced into the cement paste. Thus, tobermorite gel forms the heart of hardened cement paste and concrete in that it cements everything together.
2.2 Effects of Portland Cement Compounds
Each of the four major compounds of portland cement contributes to the behavior of the cement as it proceeds from the plastic to the hardened state after hydration. Knowledge of the behavior of each major compound upon hydration permits the amounts of each to be adjusted during manufacture to produce desired properties in the cement.
Tricalcium silicate (C3S) is primarily responsible for the high early strength of hydrated portland cement. It undergoes initial and final set within a few hours. The reaction of C3S with water gives off a large quantity of heat (heat of hydration). The rate of hardening of cement paste is directly related to the heat of hydration; the faster the set, the greater the exotherm. Hydrated C3S compound attains most of its strength in 7 days.
Dicalcium silicate (C2S) is found in three different forms, designated alpha, beta, and gamma. Since the alpha phase is unstable at room temperature and the gamma phase shows no hardening when hydrated, only the beta phase is important in portland cement.
Beta C2S takes several days to set. It is primarily responsible for the later-developing strength of
portland cement paste. Since the hydration reaction proceeds slowly, the heat of hydration is low. The beta C2S compound in portland cement generally produces little strength until after 28 days, but the final strength of this compound is equivalent to that of the C3S.
Tricalcium aluminate (C3A) exhibits an instantaneous or flash set when hydrated. It is primarily responsible for the initial set of portland cement and gives off large amounts of heat upon hydration. The gypsum added to the portland cement during grinding in the manufacturing process combines with the C3A to control the time to set. The C3A compound shows little strength increase after 1 day. Although hydrated C3A alone develops a very low strength, its presence in hydrated portland cement produces more desirable effects. An increased amount of C3A in portland cement results in faster sets and also decreases the resistance of the final product to sulfate attack.
Tetracalcium aluminoferrite (C4AF), is similar to C3A in that it hydrates rapidly and develops only low strength. Unlike C3A, however, it does not exhibit a flash set. In addition to composition, speed of hydration is affected by fineness of grinding, amount of water added, and temperatures of the constituents at the time of mixing. To achieve faster hydration, cements are ground finer. Increased initial temperature and the presence of a sufficient amount of water also speed the reaction rate.
2.3 Specifications for Portland Cements
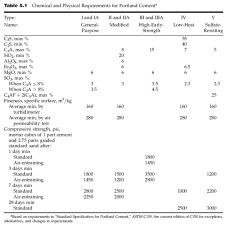
Portland cements are normally made in five types, the properties of these types being standardized on the basis of the ASTM Standard Specification for Portland Cement (C150). Distinction between the types is based on both chemical and physical requirements. Some requirements, extracted from ASTM C150, are shown in Table 5.1. Most cements exceed the strength requirements of the specification by a comfortable margin.
Type I, general-purpose cement, is the one commonly used for structural purposes when the special properties specified for the other four types of cement are not required.
Type II, modified general-purpose cement, is used where a moderate exposure to sulfate attack is anticipated or a moderate heat of hydration is required. These characteristics are attained by placing limitations on the C3A and C3S content of the cement. Type II cement gains strength a little more slowly than Type I but ultimately reaches equal strength. Type II cement, when optional
chemical requirements, as indicated in Table 5.2, are met, may be used as a low-alkali cement where alkali-reactive aggregates are present in concrete.
Type III, high-early-strength cement, is designed for use when early strength is needed in a particular construction situation. Concrete made with Type III cement develops in 7 days the same strength that it takes 28 days to develop in concretes made with Types I or II cement. This
high early strength is achieved by increasing the C3S and C3A content of the cement and by finer
grinding. No minimum is placed upon the fineness by specification, but a practical limit occurs when the particles are so small that minute amounts of moisture will prehydrate the cement during handling and storage. Since it has high heat evolution, Type III cement should not be used in large masses. With 15% C3A, it has poor sulfate resistance. The C3A content may be limited to 8% to obtain moderate sulfate resistance or to 5% when high sulfate resistance is required.
Type IV, low-heat-of-hydration cement, has been developed for mass-concrete applications. If Type I cement is used in large masses that cannot lose heat by radiation, it liberates enough heat during hydration to raise the temperature of the concrete as much as 50 or 60 8F. This results in a relatively large increase in dimensions while the concrete is still plastic, and later differential cooling after hardening causes shrinkage cracks to develop. Low heat of hydration in Type IV cement is achieved by limiting the compounds that make the greatest contribution to heat of hydration, C3A and C3S. Since these compounds also produce the early strength of cement paste, their limitation results in a paste that gains strength relatively slowly. The heat of hydration of Type IV cement usually is about 80% of that of Type II, 65% of that of Type I, and 55% of that of Type III after the first week of hydration. The percentages are slightly higher after about 1 year.
Type V, sulfate-resisting cement, is specified where there is extensive exposure to sulfates. Typical applications include hydraulic structures exposed to water with high alkali content and structures subjected to seawater exposure. The sulfate resistance of Type V cement is achieved by reducing the C3A content to a minimum since that compound is most susceptible to sulfate attack.
Types IV and V are specialty cements not normally carried in dealer’s stocks. They are usually obtainable for use on a large project if advance arrangements are made with a cement manufacturer. Air-entraining portland cements (ASTM C226) are available for the manufacture of concrete for exposure to severe frost action. These cements are available in Types I, II, and III but not in Types IV and V. When an air-entraining agent has been added to the cement by the manufacturer, the cement is designated Type IA, IIA, or IIIA.
3. Other Types of Hydraulic Cements
Although portland cements (Art. 2) are the most common modern hydraulic cements, several other kinds are in everyday use.
(to be continue..)



If you live in Weston Florida and plan on refinancing an existing mortgage or get a new mortgage beware of scam artist Tulio J. Rodriguez. This so called “Mortgage & Finance Specialist” will tell you just about anything to get your business. His group of scavengers “Real Estate Agents and Mortgage Specialists” lie through their teeth to take your money.
By: rireseets on Minggu, 2 Mei 2010
at 4:39 pm